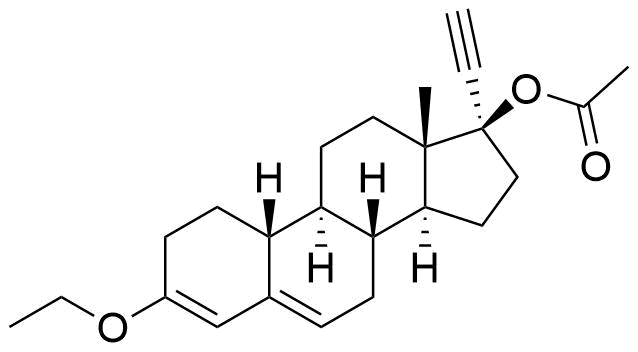
Norethindrone Acetate
Norethindrone acetate (NETA), or norethisterone acetate, is an ester prodrug of norethindrone. It is a synthetic progestogen used in place of norethindrone in some hormonal contraceptives.
Tags
Approvals
US FDA-ApprovedRelated Compounds
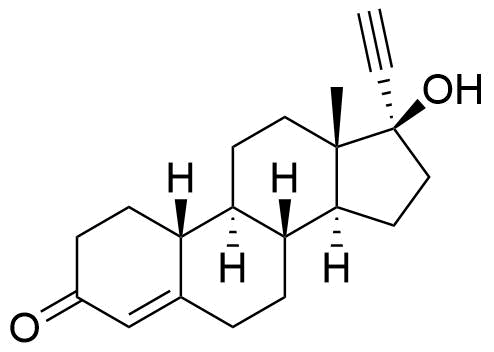
Norethindrone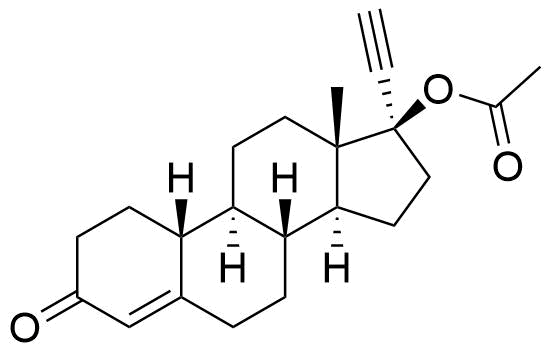
Identifiers
Abbreviation
NETA
References
Names
- norethisterone acetate
- 3-oxo-19-nor-17α-pregn-4-en-20-yn-17-yl acetate
- 17α-ethynyl-19-nortestosterone 17β-acetate
- 17α-ethynylestra-4-en-17β-ol-3-one 17β-acetate
- SH-420
References
CASRN
38673-38-0
References
PubChem CID
5832
Wikipedia Entry Name
Norethisterone Acetate
ChEBI ID
CHEBI:7628
ChEMBL ID
CHEMBL1201146
Physical & Chemical Properties
Molecular Formula
C22H28O3
References
Molecular Weight
340.5 g/mol
References
Appearance
white or yellowish-white, odorless, crystalline powder
References
- European Pharmacopoeia 8.2 Norethisterone Acetate. (View all citations for this reference)
- WHO International Agency for Research on Cancer: IARC Monographs on the Evaluation of Carcinogenic Risks to Humans. Volume 91: Combined Estrogen-Progestogen Contraceptives and Combined Estrogen-Progestogen Menopausal Therapy. 2007, Lyon, France. (View all citations for this reference)
Melting Point
161-163 °C
References
- Zafar, S.; Yousuf, S.; Kayani, H. A.; Saifullah, S.; Khan, S.; Al-Majid, A. M.; Choudhary, M. I. Biotransformation of Oral Contraceptive Ethynodiol Diacetate with Microbial and Plant Cell Cultures. Chem. Cent. J. 2012, 6 (1), 452. (View all citations for this reference)
- WHO International Agency for Research on Cancer: IARC Monographs on the Evaluation of Carcinogenic Risks to Humans. Volume 91: Combined Estrogen-Progestogen Contraceptives and Combined Estrogen-Progestogen Menopausal Therapy. 2007, Lyon, France. (View all citations for this reference)
Solubility
Practically insoluble in water (1 g in > 10 L). Soluble in chloroform (1 part in <1), ethanol (1 part in 10), dioxane (1 part in 2), and diethyl ether (1 part in 18).
References
- European Pharmacopoeia 8.2 Norethisterone Acetate. (View all citations for this reference)
- WHO International Agency for Research on Cancer: IARC Monographs on the Evaluation of Carcinogenic Risks to Humans. Volume 91: Combined Estrogen-Progestogen Contraceptives and Combined Estrogen-Progestogen Menopausal Therapy. 2007, Lyon, France. (View all citations for this reference)
Specific Optical Rotation
-32° to -28°
Polymorphism
Polymorphism identified
References
- European Pharmacopoeia 8.2 Norethisterone Acetate. (View all citations for this reference)
Toxicology
Toxicology
See Norethindrone.
References
Mutagenicity
Not found to be mutagenic in the Ames Salmonella/microsome direct plate incorporation protocol.
References
- Lang, R.; Reimann, R. Studies for a Genotoxic Potential of Some Endogenous and Exogenous Sex Steroids. I. Communication: Examination for the Induction of Gene Mutations Using the Ames Salmonella/microsome Test and the HGPRT Test in V79 Cells. Environ. Mol. Mutagen. 1993, 21 (3), 272–304. (View all citations for this reference)
Genotoxicity
Clastogenic to human lymphocytes as determined chromosome aberrations and sister chromatid exchanges. The Ames Salmonella assay did not indicate genotoxicity.
MRTD
0.0167 mg/kg/day
Biochemistry & Pharmacology
Pharmacology
See norethindrone
References
Indications
Lymphoma, hyperplasia, endometriosis.
Metabolites
Name
Structure
Notes
Norethindrone acetate, norethindrone enanthate, lynestrenol, and ethynodiol diacetate (and to a very small extent, norethynodrel) are prodrugs of norethindrone. Conversion of lynestrenol to ethynodiol to norethindrone facilitated by CYP2C9, CYP2C19, and CYP3A4. Conversion of norethynodrel to norethindrone is accomplished by either non-enzymatic or enzymatic ketosteroid isomerization.
Impurities
Name
Structure
CASRN
Other Names & Identifiers
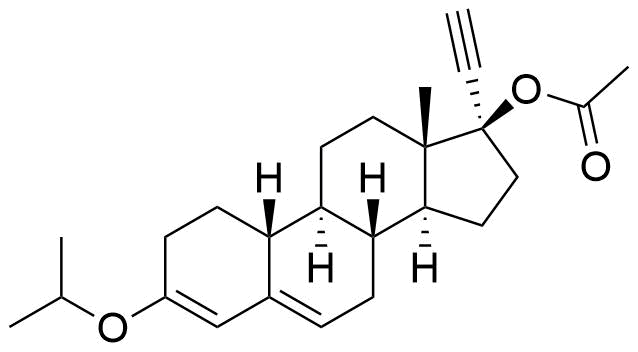
- BP Norethisterone Acetate Impurity H
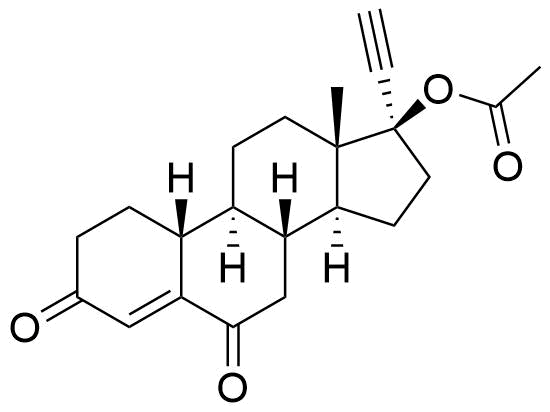
- BP Norethisterone Acetate Impurity G
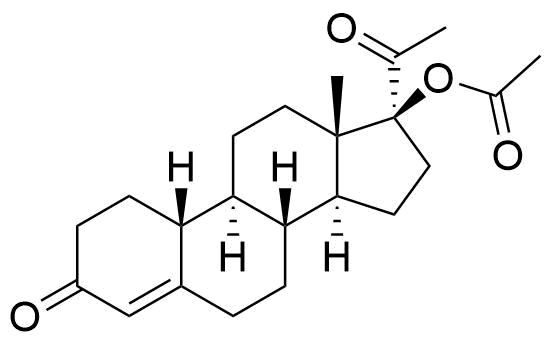
- 3,6-dioxo-19-nor-17α-pregn-4-en-20-yn-17-yl acetate
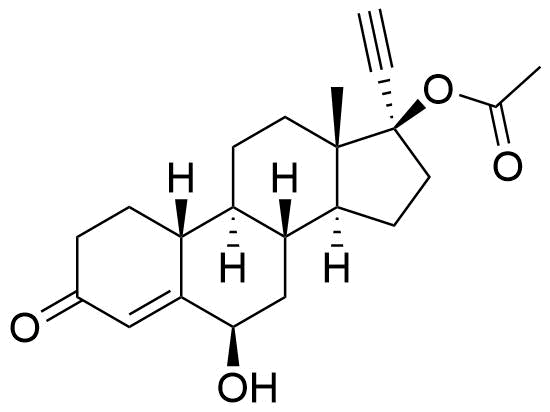
- BP Norethisterone Acetate Impurity F
- 6β-hydroxy-3-oxo-19-nor-17α-pregn-4-en-20-yn-17-yl Acetate