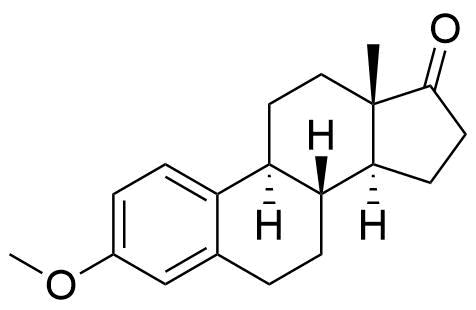
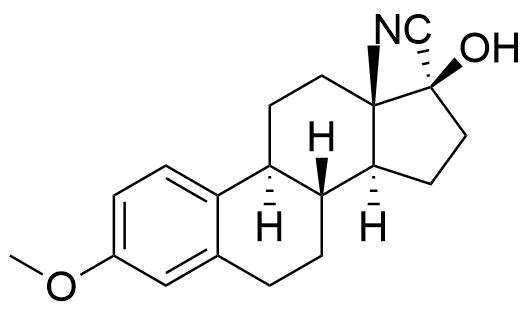
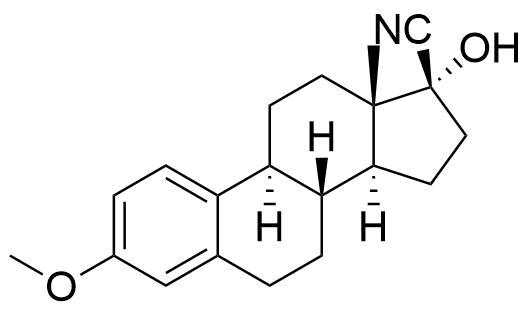
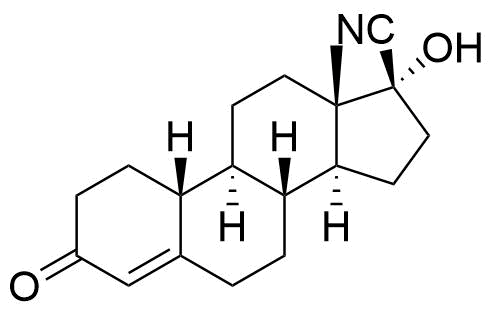
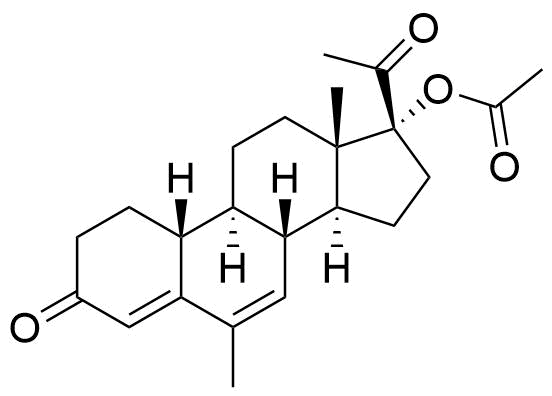
Nomegestrol Acetate
Nomegestrol acetate (NOMAC) is a synthetic progestogen used in hormonal contraceptives, hormone replacement therapy, and for the treatment of gynecological disorders. It is available internationally, but not in the US.
Tags
Identifiers
Abbreviation
NOMAC
References
Names
- 17α-acetoxy-6-methyl-19-nor-pregna-4,6-diene-3,20-dione
- 6-methyl-3,20-dioxo-19-norpregna-4,6-dien-17-yl acetate
References
CASRN
58652-20-3
References
PubChem CID
91668
ECHA InfoCard
- 100.055.781
- EC / List #: 261-379-8
UNII
83J78V5W05
KEGG Entry Number
D08281
Wikipedia Entry Name
Nomegestrol Acetate
ChEBI ID
CHEBI:135564
ChEMBL ID
CHEMBL1476022
ChemSpider ID
82771
ATC Code(s)
Physical & Chemical Properties
Molecular Formula
C23H30O4
References
Molecular Weight
370.4819 g/mol
References
Appearance
White or almost white crystalline powder
References
- British Pharmacopoeia 2017: Nomegestrol Acetate monograph (View all citations for this reference)
Melting Point
214-215° C
References
- Lello, S., Nomegestrol Acetate Pharmacology, Safety Profile and Therapeutic Efficacy. Drugs 2010, 70 (5), 541-559. (View all citations for this reference)
Solubility
Practically insoluble in water, freely soluble in acetone, soluble in ethanol (96%)
References
- British Pharmacopoeia 2017: Nomegestrol Acetate monograph (View all citations for this reference)
logP
3.46
Specific Optical Rotation
-62.6 in absolute ethanol at 22° C; -60 to -64 in absolute ethanol at 25° C
References
- Lello, S., Nomegestrol Acetate Pharmacology, Safety Profile and Therapeutic Efficacy. Drugs 2010, 70 (5), 541-559. (View all citations for this reference)
Toxicology
GHS Hazard Code(s)
| Class | Category | Code | Description |
|---|---|---|---|
| Reproductive Toxicity | 2 | H361 | Suspected of damaging fertility or the unborn child |
| Reproductive Toxicity | 1B | H360 | May damage fertility or the unborn child |
Side Effects
Abnormal bleeding/spotting, headache, hot flushes
References
- Lello, S., Nomegestrol Acetate Pharmacology, Safety Profile and Therapeutic Efficacy. Drugs 2010, 70 (5), 541-559. (View all citations for this reference)
LD50
LD50, mouse intraperitoneal: > 2 g/kg
LD50, mouse oral: > 2 g/kg
LD50, rat intraperitoneal: > 2 g/kg
LD50, rat oral: >2 g/kg
(TDLo), women oral: 1.05 mg/kg
Safety Profile Overview
Generally very well tolerated, positive safety profile
References
- Africander, D.; Verhoog, N.; Hapgood, J. P., Molecular mechanisms of steroid receptor-mediated actions by synthetic progestins used in HRT and contraception. Steroids 2011, 76 (7), 636-52. (View all citations for this reference)
- Lello, S., Nomegestrol Acetate Pharmacology, Safety Profile and Therapeutic Efficacy. Drugs 2010, 70 (5), 541-559. (View all citations for this reference)
Biochemistry & Pharmacology
Progesterone Receptor Activity
Agonist
References
Androgen Receptor Activity
Binds but no agonist activity. "Weak," "partial" or "moderate" antagonist.
References
- Africander, D.; Verhoog, N.; Hapgood, J. P., Molecular mechanisms of steroid receptor-mediated actions by synthetic progestins used in HRT and contraception. Steroids 2011, 76 (7), 636-52. (View all citations for this reference)
- Kuhl, H., Pharmacology of estrogens and progestogens: influence of different routes of administration. Climacteric 2005, 8 Suppl 1, 3-63. (View all citations for this reference)
- Su, Y.; Lian, Q. Q.; Ge, R. S., Contraceptives with novel benefits. Expert Opin Investig Drugs 2012, 21 (1), 83-90. (View all citations for this reference)
- Ruan, X.; Seeger, H.; Mueck, A. O., The pharmacology of nomegestrol acetate. Maturitas 2012, 71 (4), 345-53. (View all citations for this reference)
- Lello, S., Nomegestrol Acetate Pharmacology, Safety Profile and Therapeutic Efficacy. Drugs 2010, 70 (5), 541-559. (View all citations for this reference)
- Hapgood, J. P.; Africander, D.; Louw, R.; Ray, R. M.; Rohwer, J. M., Potency of progestogens used in hormonal therapy: toward understanding differential actions. J Steroid Biochem Mol Biol 2013, 142, 39-47. (View all citations for this reference)
- Mueck, A. O.; Sitruk-Ware, R., Nomegestrol acetate, a novel progestogen for oral contraception. Steroids 2011, 76 (6), 531-539. (View all citations for this reference)
- Shields-Botella, J.; Duc, I.; Duranti, E.; Puccio, F.; Bonnet, P.; Delansorne, R.; Paris, J., An overview of nomegestrol acetate selective receptor binding and lack of estrogenic action on hormone-dependent cancer cells. J. Steroid Biochem. Mol. Biol. 2003, 87 (2-3), 111-122. (View all citations for this reference)
Estrogen Receptor Activity
Antagonist
References
- Su, Y.; Lian, Q. Q.; Ge, R. S., Contraceptives with novel benefits. Expert Opin Investig Drugs 2012, 21 (1), 83-90. (View all citations for this reference)
- Ruan, X.; Seeger, H.; Mueck, A. O., The pharmacology of nomegestrol acetate. Maturitas 2012, 71 (4), 345-53. (View all citations for this reference)
- Lello, S., Nomegestrol Acetate Pharmacology, Safety Profile and Therapeutic Efficacy. Drugs 2010, 70 (5), 541-559. (View all citations for this reference)
- Mueck, A. O.; Sitruk-Ware, R., Nomegestrol acetate, a novel progestogen for oral contraception. Steroids 2011, 76 (6), 531-539. (View all citations for this reference)
- Shields-Botella, J.; Duc, I.; Duranti, E.; Puccio, F.; Bonnet, P.; Delansorne, R.; Paris, J., An overview of nomegestrol acetate selective receptor binding and lack of estrogenic action on hormone-dependent cancer cells. J. Steroid Biochem. Mol. Biol. 2003, 87 (2-3), 111-122. (View all citations for this reference)
Glucocorticoid Receptor Activity
No activity
References
- Kuhl, H., Pharmacology of estrogens and progestogens: influence of different routes of administration. Climacteric 2005, 8 Suppl 1, 3-63. (View all citations for this reference)
- Hapgood, J. P.; Africander, D.; Louw, R.; Ray, R. M.; Rohwer, J. M., Potency of progestogens used in hormonal therapy: toward understanding differential actions. J Steroid Biochem Mol Biol 2013, 142, 39-47. (View all citations for this reference)
- Su, Y.; Lian, Q. Q.; Ge, R. S., Contraceptives with novel benefits. Expert Opin Investig Drugs 2012, 21 (1), 83-90. (View all citations for this reference)
- Ruan, X.; Seeger, H.; Mueck, A. O., The pharmacology of nomegestrol acetate. Maturitas 2012, 71 (4), 345-53. (View all citations for this reference)
- Lello, S., Nomegestrol Acetate Pharmacology, Safety Profile and Therapeutic Efficacy. Drugs 2010, 70 (5), 541-559. (View all citations for this reference)
Mineralocorticoid Receptor Activity
No activity
References
- Kuhl, H., Pharmacology of estrogens and progestogens: influence of different routes of administration. Climacteric 2005, 8 Suppl 1, 3-63. (View all citations for this reference)
- Hapgood, J. P.; Africander, D.; Louw, R.; Ray, R. M.; Rohwer, J. M., Potency of progestogens used in hormonal therapy: toward understanding differential actions. J Steroid Biochem Mol Biol 2013, 142, 39-47. (View all citations for this reference)
- Ruan, X.; Seeger, H.; Mueck, A. O., The pharmacology of nomegestrol acetate. Maturitas 2012, 71 (4), 345-53. (View all citations for this reference)
- Lello, S., Nomegestrol Acetate Pharmacology, Safety Profile and Therapeutic Efficacy. Drugs 2010, 70 (5), 541-559. (View all citations for this reference)
Target Pathways
Bioavailability
63-65%
References
- Lello, S., Nomegestrol Acetate Pharmacology, Safety Profile and Therapeutic Efficacy. Drugs 2010, 70 (5), 541-559. (View all citations for this reference)
- Burke, A., Nomegestrol acetate-17b-estradiol for oral contraception. Patient Prefer Adherence 2013, 7, 607-19. (View all citations for this reference)
- Ruan, X.; Seeger, H.; Mueck, A. O., The pharmacology of nomegestrol acetate. Maturitas 2012, 71 (4), 345-53. (View all citations for this reference)
Elimination Half-Life (t1/2)
45-50 h
References
- Burke, A., Nomegestrol acetate-17b-estradiol for oral contraception. Patient Prefer Adherence 2013, 7, 607-19. (View all citations for this reference)
- Su, Y.; Lian, Q. Q.; Ge, R. S., Contraceptives with novel benefits. Expert Opin Investig Drugs 2012, 21 (1), 83-90. (View all citations for this reference)
- Ruan, X.; Seeger, H.; Mueck, A. O., The pharmacology of nomegestrol acetate. Maturitas 2012, 71 (4), 345-53. (View all citations for this reference)
Serum Protein Binding
98% bound to serum albumin
References
- Lello, S., Nomegestrol Acetate Pharmacology, Safety Profile and Therapeutic Efficacy. Drugs 2010, 70 (5), 541-559. (View all citations for this reference)
Metabolism
CYP3A3, CYP3A4, CYP2A6
References
- Lello, S., Nomegestrol Acetate Pharmacology, Safety Profile and Therapeutic Efficacy. Drugs 2010, 70 (5), 541-559. (View all citations for this reference)
- Nanda, K.; Stuart, G. S.; Robinson, J.; Gray, A. L.; Tepper, N. K.; Gaffield, M. E. Drug Interactions between Hormonal Contraceptives and Antiretrovirals. AIDS 2017, 31 (7), 917–952. (View all citations for this reference)
Cmax
12.3 +/- 3.5 ng/mL
References
- Burke, A., Nomegestrol acetate-17b-estradiol for oral contraception. Patient Prefer Adherence 2013, 7, 607-19. (View all citations for this reference)
- Ruan, X.; Seeger, H.; Mueck, A. O., The pharmacology of nomegestrol acetate. Maturitas 2012, 71 (4), 345-53. (View all citations for this reference)
Tmax
1.5-2 h, 2-3 h regardless of dose
References
- Lello, S., Nomegestrol Acetate Pharmacology, Safety Profile and Therapeutic Efficacy. Drugs 2010, 70 (5), 541-559. (View all citations for this reference)
- Ruan, X.; Seeger, H.; Mueck, A. O., The pharmacology of nomegestrol acetate. Maturitas 2012, 71 (4), 345-53. (View all citations for this reference)
Enzyme Interactions
Estrone sulfatase inhibitor
Metabolites
Name
Structure
Notes
6 main metabolites, all of which are basically inactive. Metabolized in the liver through hydroxylation by CYP3A3, CYP3A4, CYP2A6.