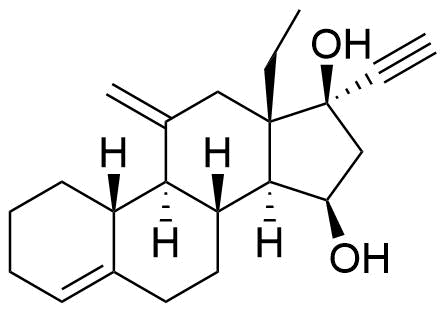
Desogestrel
Desogestrel (DSG) is a synthetic progestin and prodrug of etonogestrel (ENG), derived from testosterone and used in hormonal contraceptives. DSG is primarily used in oral contraceptives, while ENG is primarily used in implants and vaginal rings.
Tags
Approvals
WHO Prequalification US FDA-ApprovedRelated Compounds
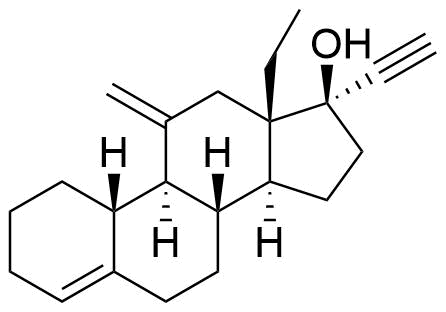
Etonogestrel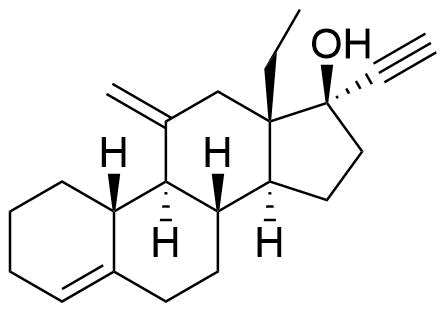
Identifiers
Abbreviation
DSG
References
Names
13-ethyl-11-methylene-18,19-dinor-17α-4- pregnen-20-yn-17-ol
References
CASRN
54024-22-5
References
PubChem CID
40973
IUPHAR/BPS
7065
KEGG Entry Number
DB00304
Wikipedia Entry Name
Desogestrel
ChEBI ID
CHEBI:4453
ChEMBL ID
CHEMBL1533
ChemSpider ID
37400
ATC Code(s)
References
- DrugBank: Desogestrel
Wishart DS, Feunang YD, Guo AC, Lo EJ, Marcu A, Grant JR, Sajed T, Johnson D, Li C, Sayeeda Z, Assempour N, Iynkkaran I, Liu Y, Maciejewski A, Gale N, Wilson A, Chin L, Cummings R, Le D, Pon A, Knox C, Wilson M. DrugBank 5.0: a major update to the DrugBank database for 2018. Nucleic Acids Res. 2017 Nov 8. doi: 10.1093/nar/gkx1037 (View all citations for this reference)
Physical & Chemical Properties
Molecular Formula
C22H30O
References
Molecular Weight
310.473 g/mol
References
Appearance
White, crystalline powder.
Melting Point
228.2-233.6 °F (109-112 °C)
References
- Desogestrel USP Safety Data Sheet. (View all citations for this reference)
- WHO International Agency for Research on Cancer: IARC Monographs on the Evaluation of Carcinogenic Risks to Humans. Volume 91: Combined Estrogen-Progestogen Contraceptives and Combined Estrogen-Progestogen Menopausal Therapy. 2007, Lyon, France. (View all citations for this reference)
Solubility
0.5 mg/L water. Freely soluble in 100% ethanol and in dichloromethane; very soluble in methanol; slightly soluble in 96% ethanol and in ethyl acetate; sparingly soluble in hexane.
References
- Toral, M. I.; Nacaratte, F.; Nova-Ramirez, F.; Otipka, R., PARALLEL DETERMINATION OF DESOGESTREL AND 17 alpha-ETHINYLESTRADIOL IN PHARMACEUTICAL FORMULATION BY DERIVATIVE SPECTROPHOTOMETRY. J. Chil. Chem. Soc. 2013, 58 (2), 1779-1784. (View all citations for this reference)
- Desogestrel USP Safety Data Sheet. (View all citations for this reference)
- WHO International Agency for Research on Cancer: IARC Monographs on the Evaluation of Carcinogenic Risks to Humans. Volume 91: Combined Estrogen-Progestogen Contraceptives and Combined Estrogen-Progestogen Menopausal Therapy. 2007, Lyon, France. (View all citations for this reference)
logP
5.6 (Toral et al., Desogestrel USP SDS)
4.27 (Desogestrel USP SDS)
References
- Toral, M. I.; Nacaratte, F.; Nova-Ramirez, F.; Otipka, R., PARALLEL DETERMINATION OF DESOGESTREL AND 17 alpha-ETHINYLESTRADIOL IN PHARMACEUTICAL FORMULATION BY DERIVATIVE SPECTROPHOTOMETRY. J. Chil. Chem. Soc. 2013, 58 (2), 1779-1784. (View all citations for this reference)
- Desogestrel USP Safety Data Sheet. (View all citations for this reference)
Specific Optical Rotation
[α]20D, +53° to -57° in chloroform.
Storage Conditions
Stable at normal conditions.
Toxicology
Side Effects
General progestin side effects: Abnormal uterine bleeding; breast enlargement, pain, or tenderness; unexpected or increased flow of breast milk; hot flashes; loss of sexual desires; dry mouth; unusual thirst; frequent urination; abdominal pain; cramping; nausea; loss of appetite; dizziness; headache; mood changes; insomnia; unusual tiredness or weakness; swelling in ankles or feet; unusual or rapid weight gain; skin rash; acne; loss or gain of hair; brown spots on skin; sudden changes in vision.
Carcinogenicity
No treatment-related tumorigenic effects seen from administration of desogestrel only. Increase in mammary adenocarcinoma in female rats in the high-dose group compared to controls.
Mutagenicity
Not associated with point mutations in the in vitro Ames test and to chromosomal aberrations in a micronucleus test in female rats.
LD50
- Mouse oral: > 2000 mg/kg
- Rat oral: > 2000 mg/kg
MRTD
0.0025 mg/kg/day
Biochemistry & Pharmacology
Progesterone Receptor Activity
Agonist
References
- DrugBank: Desogestrel
Wishart DS, Feunang YD, Guo AC, Lo EJ, Marcu A, Grant JR, Sajed T, Johnson D, Li C, Sayeeda Z, Assempour N, Iynkkaran I, Liu Y, Maciejewski A, Gale N, Wilson A, Chin L, Cummings R, Le D, Pon A, Knox C, Wilson M. DrugBank 5.0: a major update to the DrugBank database for 2018. Nucleic Acids Res. 2017 Nov 8. doi: 10.1093/nar/gkx1037 (View all citations for this reference)
Estrogen Receptor Activity
ERα: Agonist
References
- DrugBank: Desogestrel
Wishart DS, Feunang YD, Guo AC, Lo EJ, Marcu A, Grant JR, Sajed T, Johnson D, Li C, Sayeeda Z, Assempour N, Iynkkaran I, Liu Y, Maciejewski A, Gale N, Wilson A, Chin L, Cummings R, Le D, Pon A, Knox C, Wilson M. DrugBank 5.0: a major update to the DrugBank database for 2018. Nucleic Acids Res. 2017 Nov 8. doi: 10.1093/nar/gkx1037 (View all citations for this reference)
Target Pathways
Bioavailability
76% as etonogestrel.
References
- Grandi, G.; Cagnacci, A.; Volpe, A. Pharmacokinetic Evaluation of Desogestrel as a Female Contraceptive. Expert Opin. Drug Metab. Toxicol. 2014, 10 (1), 1–10. (View all citations for this reference)
- Schindler, A. E.; Campagnoli, C.; Druckmann, R.; Huber, J.; Pasqualini, J. R.; Schweppe, K. W.; Thijssen, J. H. H., Classification and pharmacology of progestins. Maturitas 2003, 46, 7-16. (View all citations for this reference)
Elimination Half-Life (t1/2)
30 h (single or multiple doses)
Serum Protein Binding
As etonogestrel: 58% to serum albumin, 38% to SHBG, 3.5% unbound. (Grandi)
32% to SHBG, 66% to albumin, 2% free. (Schindler)
References
- Grandi, G.; Cagnacci, A.; Volpe, A. Pharmacokinetic Evaluation of Desogestrel as a Female Contraceptive. Expert Opin. Drug Metab. Toxicol. 2014, 10 (1), 1–10. (View all citations for this reference)
- Schindler, A. E.; Campagnoli, C.; Druckmann, R.; Huber, J.; Pasqualini, J. R.; Schweppe, K. W.; Thijssen, J. H. H., Classification and pharmacology of progestins. Maturitas 2003, 46, 7-16. (View all citations for this reference)
Metabolism
CYP3A4, CYP2C9
Excretion
60% excreted via urine, 35% excreted via feces.
Cmax
- 640 pg/mL from oral administration of 75 μg desogestrel
- 4273 +/- 830 ng/L from 30 μg ethinyl estradiol/150 μg desogestrel oral pill
Tmax
< 2 h from multiple oral administration of 75 μg desogestrel (Grandi)
1.15 h (Schindler)
References
- Grandi, G.; Cagnacci, A.; Volpe, A. Pharmacokinetic Evaluation of Desogestrel as a Female Contraceptive. Expert Opin. Drug Metab. Toxicol. 2014, 10 (1), 1–10. (View all citations for this reference)
- Schindler, A. E.; Campagnoli, C.; Druckmann, R.; Huber, J.; Pasqualini, J. R.; Schweppe, K. W.; Thijssen, J. H. H., Classification and pharmacology of progestins. Maturitas 2003, 46, 7-16. (View all citations for this reference)
Inhibition of Ovulation
0.06 mg/day
References
- Rebar, R. W.; Zeserson, K., CHARACTERISTICS OF THE NEW PROGESTOGENS IN COMBINATION ORAL-CONTRACEPTIVES. Contraception 1991, 44 (1), 1-10. (View all citations for this reference)
Transformation of Endometrium
2.5 mg/cycle
References
- Rebar, R. W.; Zeserson, K., CHARACTERISTICS OF THE NEW PROGESTOGENS IN COMBINATION ORAL-CONTRACEPTIVES. Contraception 1991, 44 (1), 1-10. (View all citations for this reference)
Menstrual Delay
~0.25 (mg/day
References
- Rebar, R. W.; Zeserson, K., CHARACTERISTICS OF THE NEW PROGESTOGENS IN COMBINATION ORAL-CONTRACEPTIVES. Contraception 1991, 44 (1), 1-10. (View all citations for this reference)
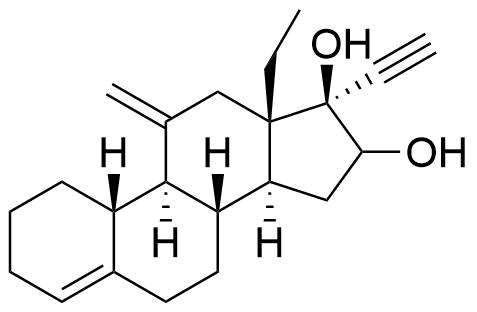
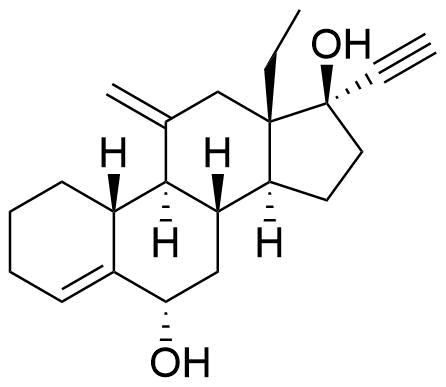
Metabolites
Name
Structure
Notes
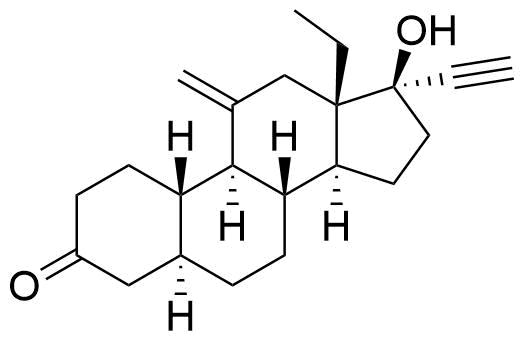
Lower Progesterone Receptor and Estrogen Receptor activity than etonogestrel.
Glucuronide and sulfate conjugates of desogestrel and some of its metabolites (3β-hydroxydesogestrel, 15β-hydroxydesogestrel) are also formed.
Impurities
Name
Structure
CASRN
Other Names & Identifiers
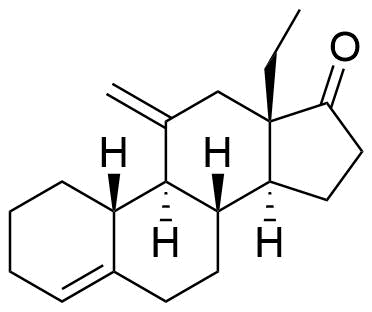
54024-21-4
- USP Desogestrel Related Compound D
- BP Desogestrel Impurity C
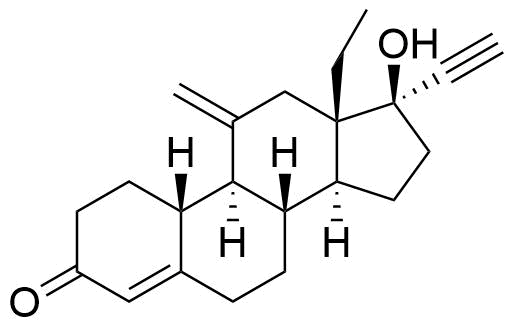
54048-10-1
- USP Desogestrel Related Compound C
- BP Desogestrel Impurity D
- 13-Ethyl-11-methylene-18,19-dinor-17α-pregn-4-en-20-yn-17-ol-3-one
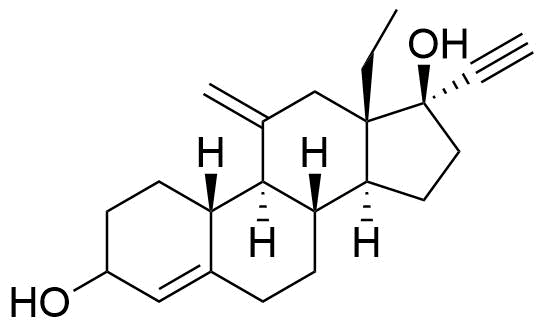
70805-85-5
- USP Desogestrel Related Compound B
- BP Desogestrel Impurity E
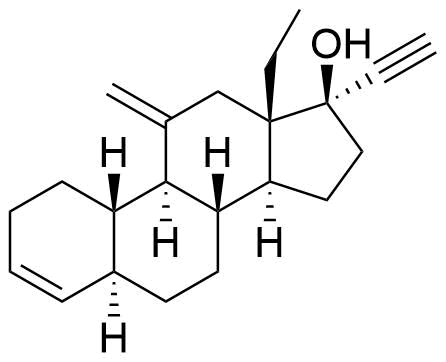
201360-82-9
- USP Desogestrel Related Compound A
- BP Desogestrel Impurity A
- Desogestrel Δ3 Isomer
US FDA-Approved Products
Name
Formulation
Status
ANDA #
ETHINYL ESTRADIOL: 0.02 mg, 0.01 mg
28 Oral Tablets
Prescription
209170
Prescription
021090
Prescription
202689
Prescription
091346
WHO Prequalified Medicines
WHO Reference #
Name
Applicant
Formulation
ETHINYL ESTRADIOL: 30 μg
Tablet
Placebo tablet: 0 mg
ETHINYL ESTRADIOL: 30 μg
Tablet
Placebo tablet: 0 mg
ETHINYL ESTRADIOL: 0.030 mg
Tablet
ETHINYL ESTRADIOL: 0.030 mg
Placebo: 0 mg
Tablet