Norethindrone
Structure
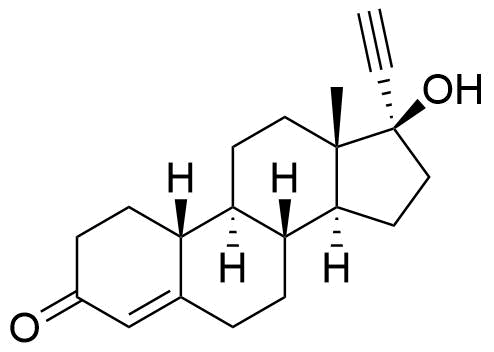
Other Names & Identifiers
- BP Norethisterone Acetate Impurity A
- Norethisterone
CASRN
68-22-4
Limits
0.1%
References
- British Pharmacopoeia 2014: Norethisterone API (View all citations for this reference)
- European Pharmacopoeia 8.2 Norethisterone (View all citations for this reference)
- British Pharmacopoeia 2017: Norethisterone monograph. (View all citations for this reference)
- Gorog SH, B. Analysis of Steroids. 38. The use of high-performance liquid chromatography with diode-array UV detection for estimating impurity profiles of steroid drugs. Journal of Chromatography. 1987; 400:177-86. (View all citations for this reference)
- Babják, M.; Balogh, G.; Gazdag, M.; Görög, S. Estimation of Impurity Profiles of Drugs and Related Materials: Part XXI. HPLC/UV/MS Study of the Impurity Profile of Ethynodiol Diacetate. J. Pharm. Biomed. Anal. 2002, 29 (6), 1153–1157. (View all citations for this reference)